(Carl) Bosch
Carl Bosch ( 1874 - 1940)

Carl Bosch, born in Cologne, Germany, was a prominent German industrial chemist and entrepreneur.
Carl Bosch’s uncle was the legendary industrialist Robert Bosch who helped develop the first spark plug.
Carl himself developed the Haber-Bosch process for high-pressure synthesis of ammonia and was one of the founders of IG Farben, which became one of the world’s largest chemical companies.
1931 Bosch won the Nobel Prize for Chemistry for formulating chemical high-pressure methods, the first Nobel in chemistry given for practical rather than theoretical discoveries.
Bosch began his studies in mechanical engineering and metallurgy, the study of metallic elements in 1894 and within two years began to study chemistry and received his doctorate in 1898.
After graduating, Bosch was given his first job as a chemist at Badische Anilin- and Soda-Fabrik.
They recruited Bosch to create a synthetic indigo color (dye) for cotton, the color used for blue jeans.
While working at BASF in 1908, Bosch met Fritz Haber who had been working on synthesizing ammonia from hydrogen and atmospheric nitrogen (One of the primary chemicals in fertilizer that makes plants grow).
What exactly is the Haber-Bosch process?
At the end of the nineteenth century, supplying the expanding population with food, raised a growing difficulty, since the nitrogen content of the soils exhausted, whereas the industrial support of nitrogen fertilizers remained unsolved.
Nitrogen is primarily assimilated in the form of nitrate ions (is an atom or molecule in which the total number of electrons is not equal to the number of protons, giving the atom a net positive or negative electric charge) and ammonium ions by plants, it's very common in the atmosphere, but difficult to get into a liquid or solid form that can be used. Doing so is known as “fixing” nitrogen.
At the beginning of the twentieth century scientists had been trying for more than 100 years to fix or synthesize nitrogen in a man-made manner.
In 1908 Fritz Haber discovered the principles of ammonia synthesis, utilizing all the physical and chemical resources available that time. The synthesis takes place in the presence of an iron catalyst, at high pressure and temperature.
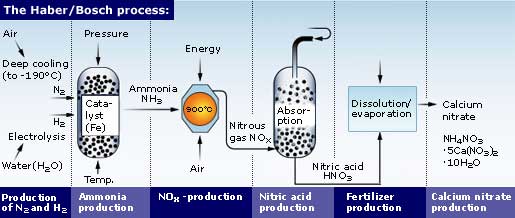
Carl Bosch commercialized the process by establishing the first industrial-level production in 1913.
This industrial process allowed the expansion of both, agriculture production and human population, during the 20th century and so created a capacity to sustain the world's need for food production.
"The Haber-Bosch-Process has been of greater fundamental importance to the modern world than ... the airplane, nuclear energy, space flight, or television. The expansion of the world's population from 1.6 billion people in 1900 to six billion in 2000 would not have been possible without the synthesis of ammonia." - Vaclav Smil (Canadian scientist and policy analyst).
Famous Chemist
- (Gertrude) B. Elion
- (Louis) Lumière
- (Carl) Bosch
- (John) Dalton
- (Antoine Laurant) de Lavoisier
- (Dimitri) Mendelejew
- (Alfred) Nobel
- (Ernest) Rutherford
 Gertrude B. Elion (1918-1999)
Gertrude B. Elion (1918-1999)
Gertrude B. Elion was born to immigrant parents in New York city. Her father came from Lithuania and her mother from Russia. She spent her early years in Manhattan where her father had a dental practice. When her brother was born they moved to Bronx.
Louis Lumière (1864-1948)

Nobody of us can imagine the world without movies, cinemas and actors. It is unbelievable especially for people who are living in XX century. But you have to know that it was not always like this. The possibility to go to the cinema and watch a new movie gave us Louis Lumière and his brother Auguste.
Read more...Carl Bosch ( 1874 - 1940)

Carl Bosch, born in Cologne, Germany, was a prominent German industrial chemist and entrepreneur.
Read more... John Dalton (1766- 1844)
John Dalton (1766- 1844)
He is a famous English naturalist and teacher. He is considered as an innovator of chemistry because of his fundamental researches. Here are some examples of his successful research: the studies about thermal expansion, formulates the "Dalton- law", preparatory work of the periodic system of elements and he discovered the "Daltonism" which means acritochromacy.
 Antoine Laurant de Lavoisier (1743-1794)
Antoine Laurant de Lavoisier (1743-1794)
In 1772 he discovered the three conditions of matters: solid, liquid and gassy which he published in "Opuscule physique et chemique" (franc., small discourse of chemistry and physics). Additionally, he gave oxygen (acid-creator) his name because he discovered while some experiments that water and air are compound substances. Meanwhile the French Revolution, Lavoisier helped in introducing the "metric system" which unifies all of units.
 Dimitri Mendelejew (1834- 1907)
Dimitri Mendelejew (1834- 1907)
has succeeded to tabulate the periodic system (short: PSE= periodic system of elements) which contains every chemical element. He started the system to impose beginning with the smallest to the biggest element. When there were any properties in common, he put them into groups and in the periodic system he arranged them among themselves. In the end, Mendelejew illustrated a context between atomic weight and chemical characteristics. The element 101 "Mendelevium" was called by Mendelejew.
 Alfred Nobel (1833- 1896)
Alfred Nobel (1833- 1896)
affected by his father and his engineering works, Nobel's career begins with some smaller inventions (gas -and liquid measuring instrument, barometer or manometer) which he also patented. In May 1862, Nobel ran the first successful underwater demolition with nitroglycerin. Two years later, he invented the detonation which he has called "Nobel's patent detonator". His idea of quality manufacturing was not safe to operate because a dynamite factory exploded -and even Nobel's little brother died. But Nobel did not surrender, he went on in improving the detonations.
 Ernest Rutherford (1871-1937)
Ernest Rutherford (1871-1937)
since 1931 was an atomic physicist. Until he was awarded with the Nobel price in 1908, Rutherford made some discoveries of highest priority for chemists. One example could be the half life before he has been honored because of his studies about the decay of elements and the chemical composition of radioactive elements. In the year 1911, Rutherford refutes the atomic model of Joseph J. Thomson and originates the "Rutherfordium atomic model". Additionally, the first artificial nuclear reaction was also succeeded by Rutherford.








